The Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP) provide guidance on the safe use of vaccines and related biological products, including post-exposure prophylaxis (PEP). When administered according to ACIP guidelines, including thoroughly infiltrating all wounds with human rabies immune globulin (HRIG), PEP is essentially 100% effective in preventing human rabies.1
The Following Videos Provide Specific Guidance on How to Properly Administer KEDRAB® (Rabies Immune Globulin [Human]):
KEDRAB Administration Video

This animated video provides step-by-step visual guidance on how to properly dose and administer KEDRAB to previously unvaccinated patients – for immediate protection from the rabies virus. It also includes details on the 3 crucial steps of PEP (according to the CDC–ACIP), common reasons for PEP failure, and the importance of proper HRIG volume during administration.
Real-World HRIG Administration Video
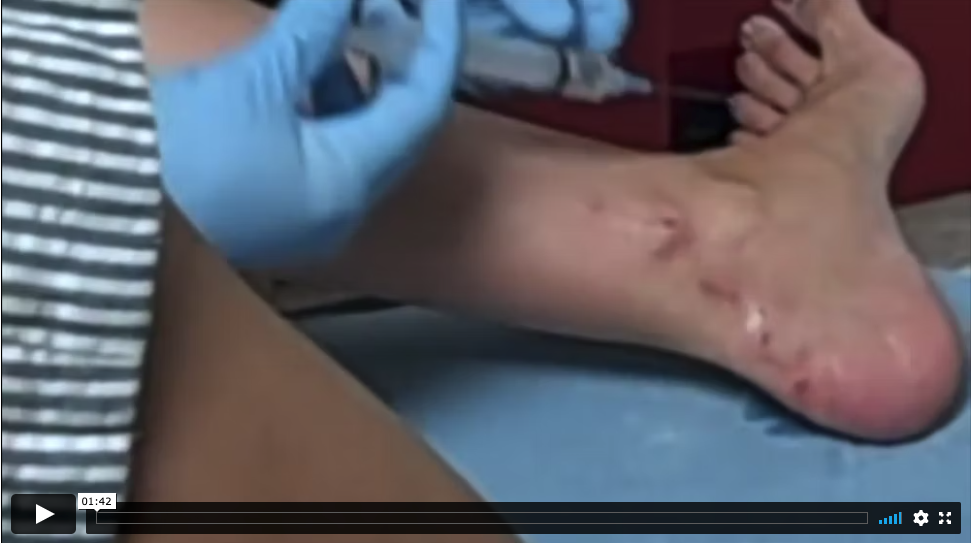
View an actual patient being administered HRIG in the emergency department as part of PEP after a potential rabies exposure from a fox bite. The healthcare provider properly advises the patient that he needs to administer as much of the HRIG dose as possible into and around the area of the exposure.
HRIG Volume Matters
References: 1. Centers for Disease Control and Prevention. Human rabies prevention—United States, 2008: recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2008;57(RR-3):1-28. 2. KEDRAB [package insert]. Fort Lee, NJ: Kedrion Biopharma Inc.; 2021. 3. Bookstaver PB, Akpunonu P, Nguyen HB, Swan JT, Howington GT. Administration of rabies immunoglobulin: Improving evidence-based guidance for wound infiltration. Pharmacotherapy. 2021;41(8):644-648.
